 
Selected values are provided in the table below. When A=0, the activity coefficients are unity, thus describing an ideal mixture.Īn extensive range of recommended values for the Van Laar coefficients can be found in the literature. In this case the activity coefficients mirror at x 1=0.5. 
The equation approximates the behavior of real. ) It was derived by Johannes Diderik van der Waals in 1873, based on a modification of the ideal gas law. Van Laar derived the excess enthalpy from the van der Waals equation: H e x = b 1 X 1 b 2 X 2 b 1 X 1 + b 2 X 2 ( a 1 b 1 − a 2 b 2 ) 2 The van der Waals equation is an equation of state for a fluid composed of particles that have a non-zero size and a pairwise attractive inter-particle force (such as the van der Waals force. Because of this, the model lost the connection to molecular properties, and therefore it has to be regarded as an empirical model to correlate experimental results. The original van der Waals parameters didn't give good description of vapor-liquid equilibria of phases, which forced the user to fit the parameters to experimental results. The equation was derived from the Van der Waals equation. The Van Laar equation is a thermodynamic activity model, which was developed by Johannes van Laar in 1910-1913, to describe phase equilibria of liquid mixtures. Air - Molecular Weight and Composition - Dry air is a mixture of gases where the average molecular weight (or molar mass) can be calculated by adding the weight of each component.Mathematical model of the thermodynamic activity of phase equilibria of liquid mixtures.Gases and Compressed Air - Air, LNG, LPG and other common gas properties, pipeline capacities, sizing of relief valves.Involving velocity, pressure, density and temperature as functions of space and time. By adding corrections for interparticle attractions and particle volumes to the ideal gas law, we can derive a new equation that more accurately describes real gas behavior. Fluid Mechanics - The study of fluids - liquids and gases.Air Psychrometrics - Moist and humid air - psychrometric charts, Mollier diagrams, air-condition temperatures and absolute and relative humidity and moisture content.Basics - The SI-system, unit converters, physical constants, drawing scales and more.* To convert a into kPa L2/mol2 multiply by 100.0 kPa/bar * To convert a into atm L2/mol2 multiply by 0.986 atm/bar To convert a into kPa L 2/mol 2 multiply by 100.0 kPa/bar Name 
* To convert a into atm L 2/mol 2 multiply by 0.986 atm/bar. How to calculate total pressure and partial pressures from Ideal gas law See also the Gas Compressibility Factor, z-factor for air, dependent on pressure and temperature and used to account for deviation from the ideal situation. 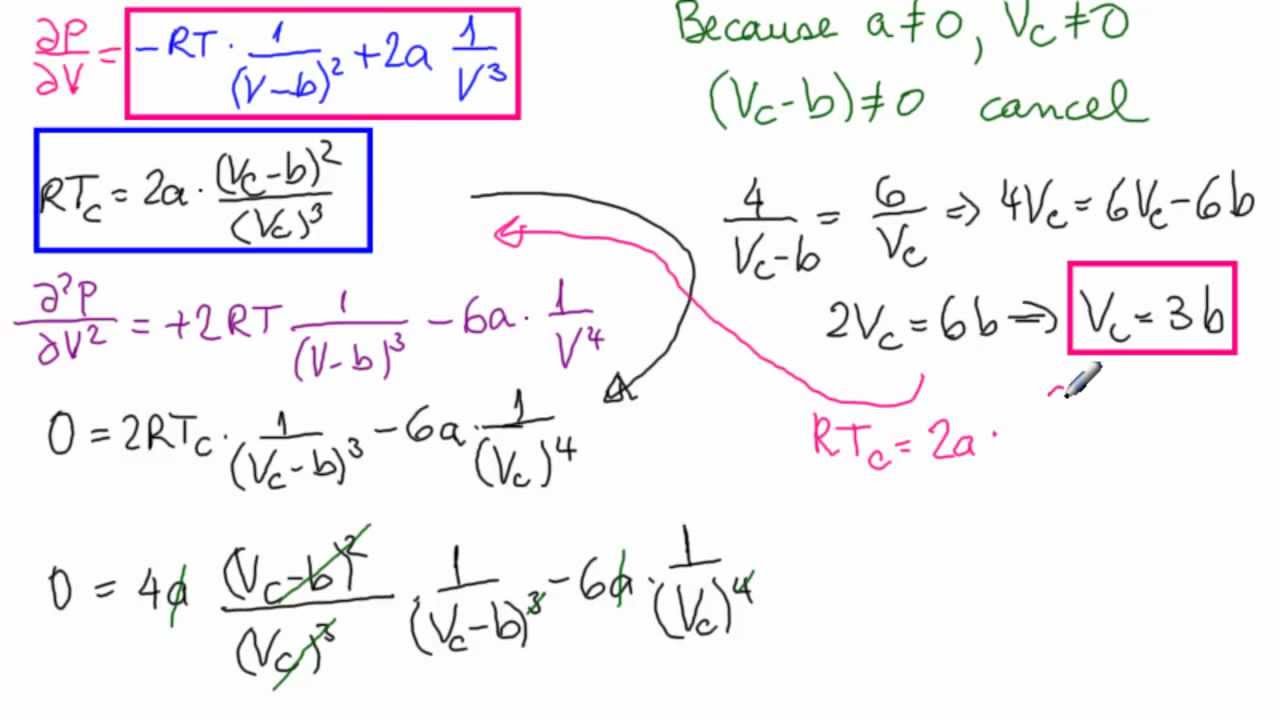
It is a correction for finite molecular size and its value is the volume of one mole of the atoms or molecules. The constant a provides a correction for the intermolecular forces.Ĭonstant b adjusts for the volume occupied by the gas particles. If a gas behaves ideally, both a and b are zero, and van der Waals equations approaches the ideal gas law PV=nRT. They have positive values and are characteristic of the individual gas. The constants a and b are called van der Waals constants. It is usually referred to as the van der Waals equation of state: van der Waals suggested a modification to take into account molecular size and molecular interaction forces. However, gas molecules are not point masses, and there are many cases gases need to be treated as non-ideal. This works well for dilute gases in many experimental circumstances. The ideal gas law treats the molecules of a gas as point particles with perfectly elastic collisions. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
